Summary
DeepMind’s latest AlphaFold model has made significant strides in drug discovery by accurately predicting the structures of proteins, nucleic acids, and ligands. This article delves into the capabilities of this game-changing technology, its application in therapeutic drug design, and its limitations.
Photo from deepmind.google
Five years ago, DeepMind, a leading AI research lab under Google, introduced the world to AlphaFold, an artificial intelligence system capable of predicting the structures of numerous proteins within the human body. As of today, the pioneering technology has evolved into AlphaFold 2 and beyond, demonstrating unprecedented predictive capabilities that are revolutionizing the world of drug discovery.
“The potential of AI to greatly enhance scientific understanding of the molecular machines that make up the human body.”
Unveiling the New AlphaFold
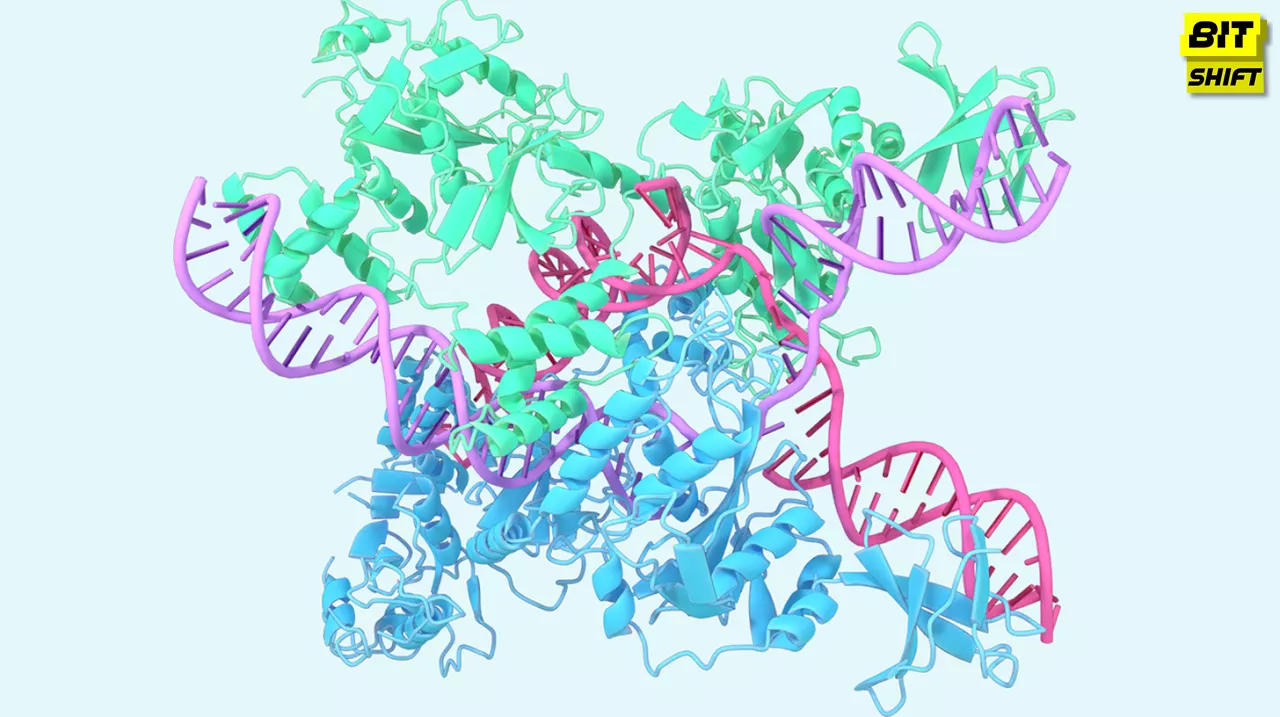
DeepMind recently revealed that the latest iteration of AlphaFold can generate predictions for nearly all molecules in the Protein Data Bank, the world’s most extensive open-access database of biological molecules. The new AlphaFold model, co-designed by DeepMind and its spin-off, Isomorphic Labs, is already being applied to therapeutic drug design. Per a post on DeepMind’s blog, the model is helping characterize various molecular structures vital for treating diseases.
The Expanded Capabilities of AlphaFold
The new AlphaFold’s competencies extend beyond mere protein prediction. According to DeepMind, the model can accurately predict the structures of ligands, molecules that bind to “receptor” proteins and incite changes in cellular communication, and nucleic acids, molecules containing essential genetic information. It can also predict post-translational modifications, which are chemical changes after a protein’s creation.
DeepMind asserts that predicting protein-ligand structures can significantly aid drug discovery, enabling scientists to identify and design new molecules that could become drugs. Until now, pharmaceutical researchers relied on computer simulations known as “docking methods” to determine how proteins and ligands would interact. This necessitated specifying a reference protein structure and a suggested binding position for the ligand. However, the latest AlphaFold eradicates the need for these steps. The model can predict proteins that haven’t been “structurally characterized” before while simultaneously simulating how proteins and nucleic acids interact with other molecules, a modeling level that DeepMind claims is unachievable with today’s docking methods.
The Road Ahead and Current Limitations
Despite its significant advancements, the newest AlphaFold has its limitations. In a whitepaper detailing the system’s strengths and weaknesses, researchers from DeepMind and Isomorphic Labs explain that the system needs to be a better-in-class method for predicting the structures of RNA molecules, which carry the instructions for protein creation in the body. However, DeepMind and Isomorphic Labs are dedicated to addressing this shortfall and optimizing the system’s performance.
The latest developments in AlphaFold demonstrate the vast potential of AI in enhancing our understanding of the human body’s molecular machinery. With further advancements, we can anticipate a future where AI will play an even more central role in drug discovery and other medical breakthroughs.